Closeout Procedures
Categories:
Introduction
Closeout Procedures
Closeout is a post award activity that officially ends the award relationship. It is the process in which the awarding agency determines that all applicable administrative actions and all required work with the award have been completed by the recipient.
Effective research administration over the life of the award will help eliminate problems at closeout. Manage the project closely during the award:
-
Keep faculty and staff informed of the rules not just during the 90 days after the end of the award.
-
Set up early and extended PTAs.
-
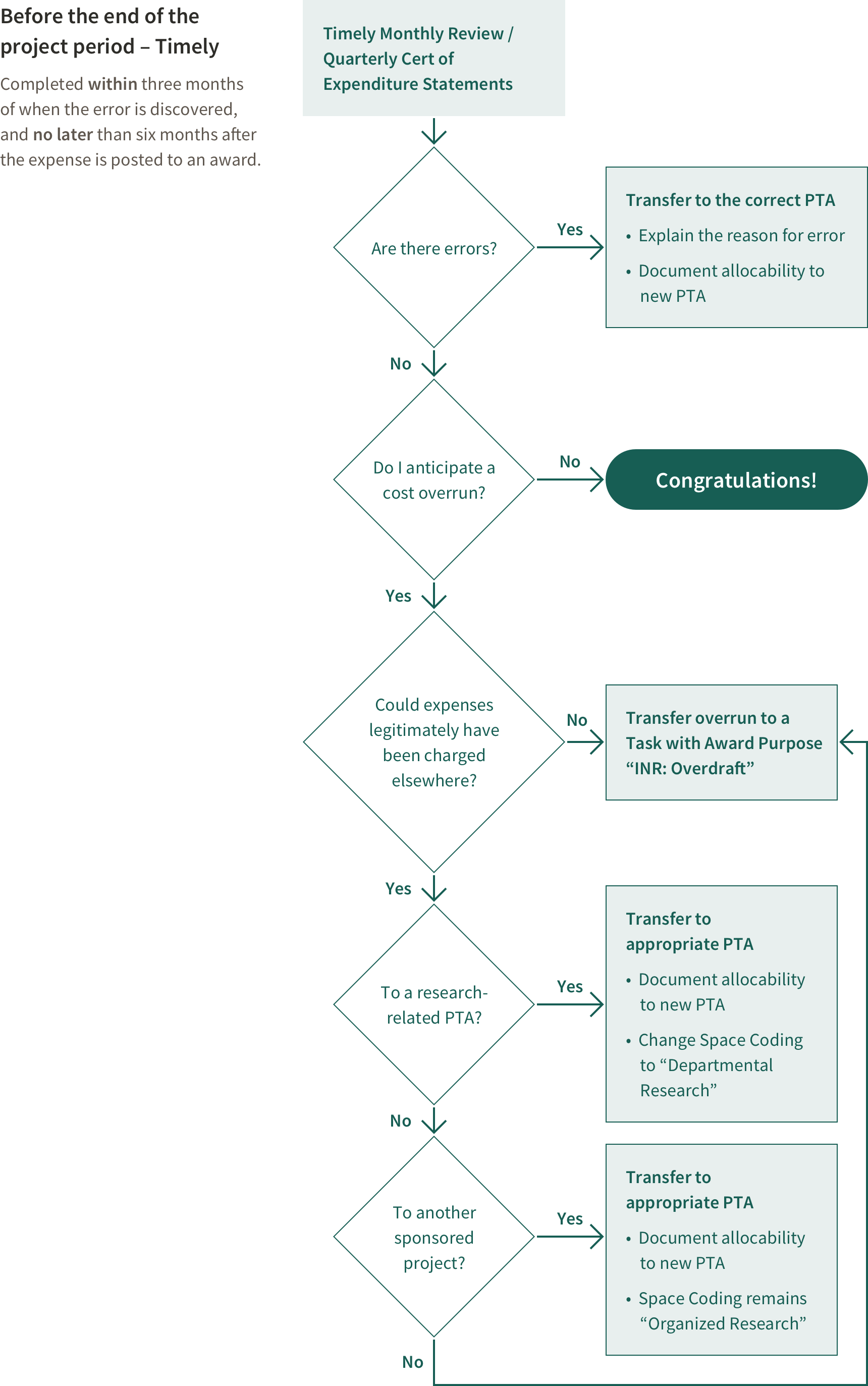
Review PTAs regularly to avoid last minute transfers. All expenses must post to the PTA within 60 days of the project end date.
Sponsor Requirements
Federal sponsors have a requirement that closeout documents be submitted within 90 days after the end of the period of performance; therefore, the research administrator must ensure that expenses are complete and have posted to the PTA within 60 days of the project end date.
-
The Office of Sponsored Research (OSR) reviews sponsored projects at closeout to ensure that costs are allowable, allocable, reasonable, and consistent. OSR then prepares the final financial report and submits it to the sponsor.
-
Sponsors have up to four years after the official closeout date to conduct a closeout audit.
-
It is critical for the PI to submit all technical and invention reports that are due.
Report Requirements
During closeout Stanford must submit all required financial, performance, and other reports as required by the award, and liquidate all obligations incurred under the award. Sponsors must make prompt payments for allowable, reimbursable grant costs. The award notice specifies the closeout report requirements.
The Technical Report should include:
-
Grant number, PI's name, project title, and performance period, including authorized extensions
-
Significant results of the project
-
Examples of progress
-
Technical difficulties and solutions
-
List of publications, including articles in progress
-
If the award contained objectives and deliverables, does my report discuss each of these?
Financial Report
OSR will prepare the final financial report and submit it to the sponsor. Sponsors have up to four years after the official closeout date to conduct a closeout audit. All financial records should be retained for a minimum of four years after closeout, longer if circumstances require it. The four-year retention period can be expanded in the case of lawsuits, patent applications, charges of misconduct, conflict of interest, etc.
Have all project expenditures been posted, i.e., have they appeared on monthly expenditure statements?
Are all project expenditure statements certified? Is the summary of expenditures (for NIH awards) certified?
Invention Report
The report must include:
-
Name of the contractor and contract number
-
Name of inventor(s)
-
Title of invention(s)
-
Patent or disclosure number
-
Data produced under the agreement
-
Reporting period
-
Royalty income if any
Property Report
Identify all materials and equipment acquired under this award, including property that was:
-
Provided by the sponsor
-
Acquired by my department
-
Purchased by a subcontractor
Equipment records must be kept for four years after final disposition of equipment or four years after project closeout, whichever is later.
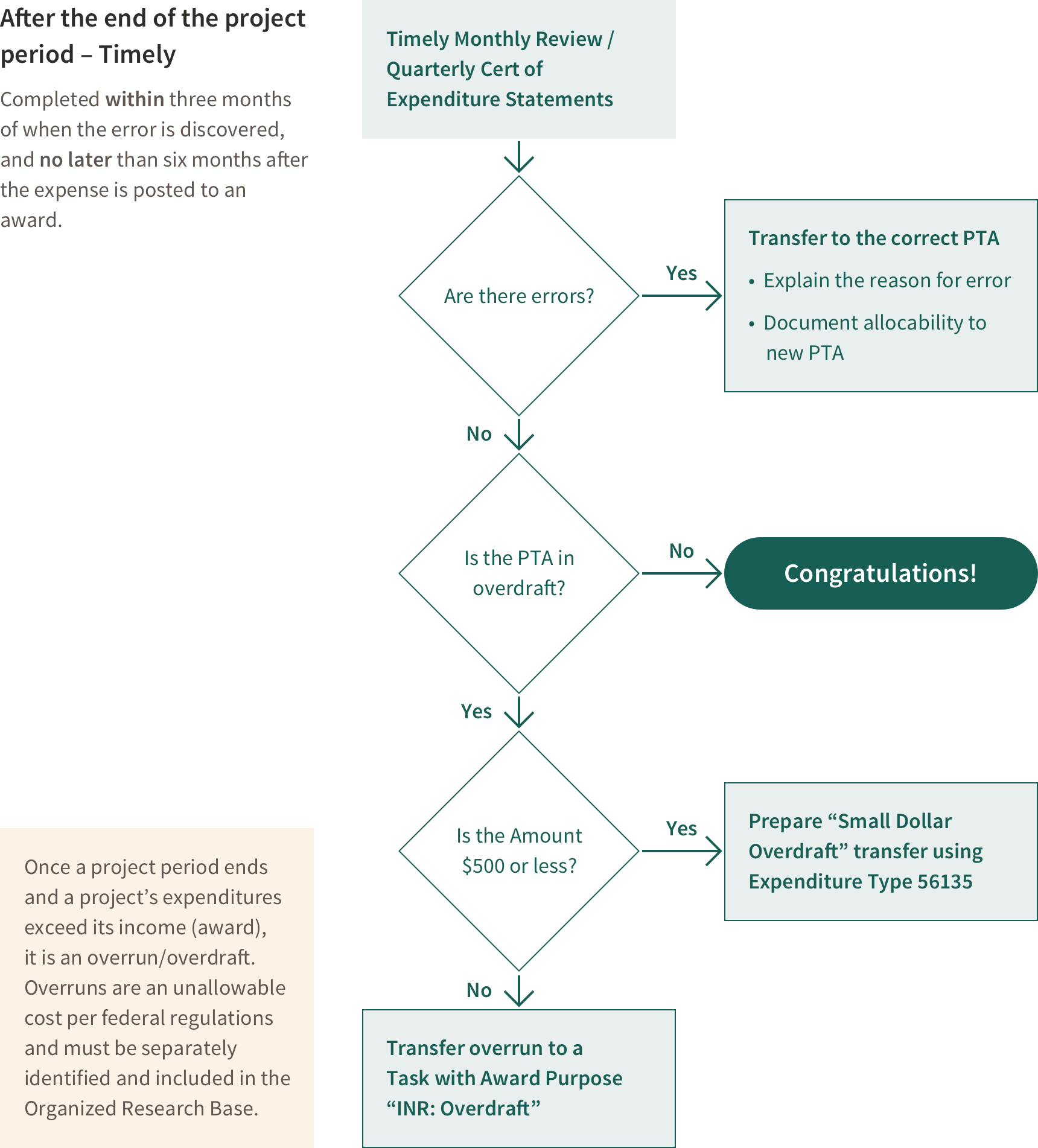
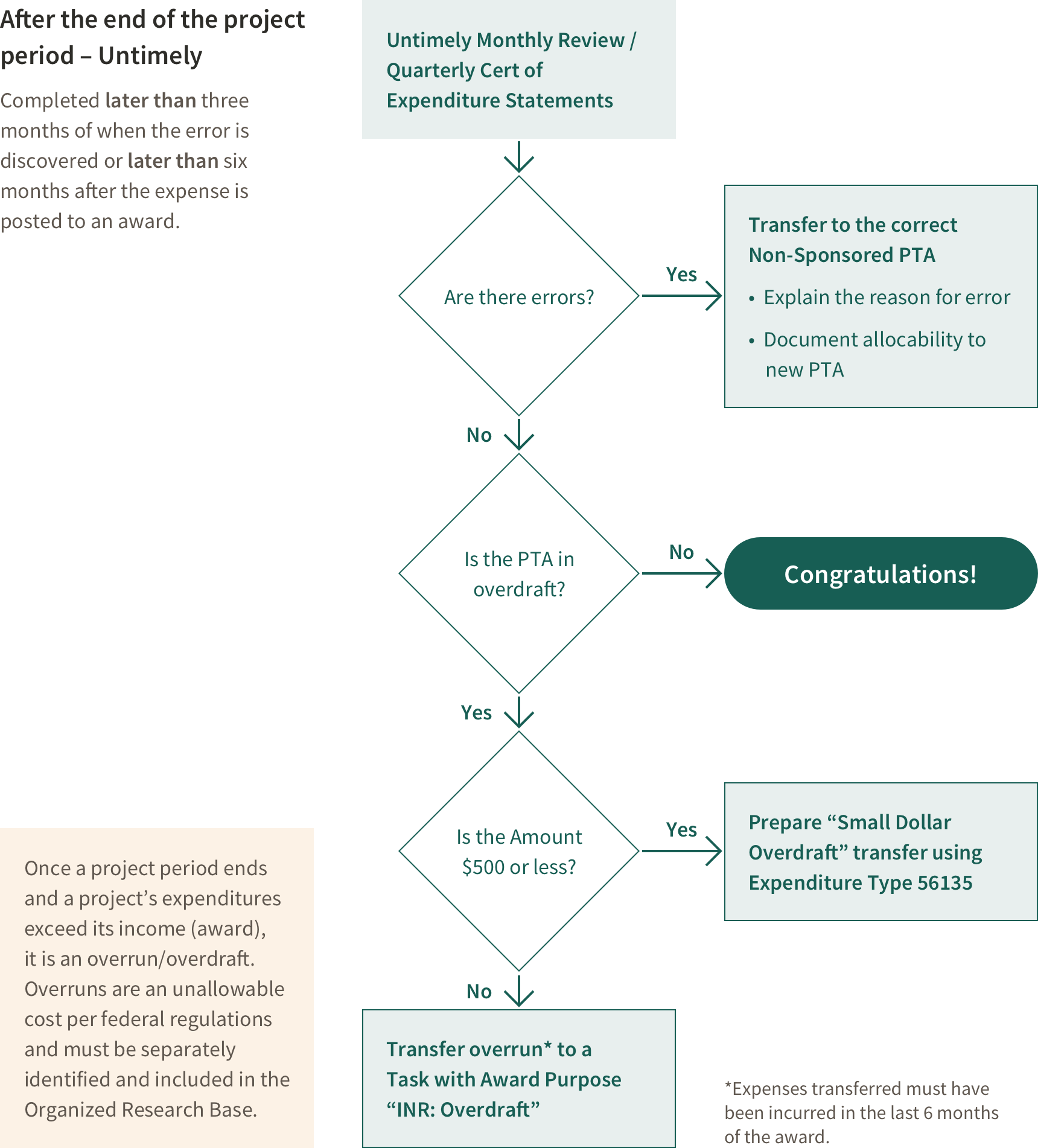
Clearing an Overdraft After the Award End Date
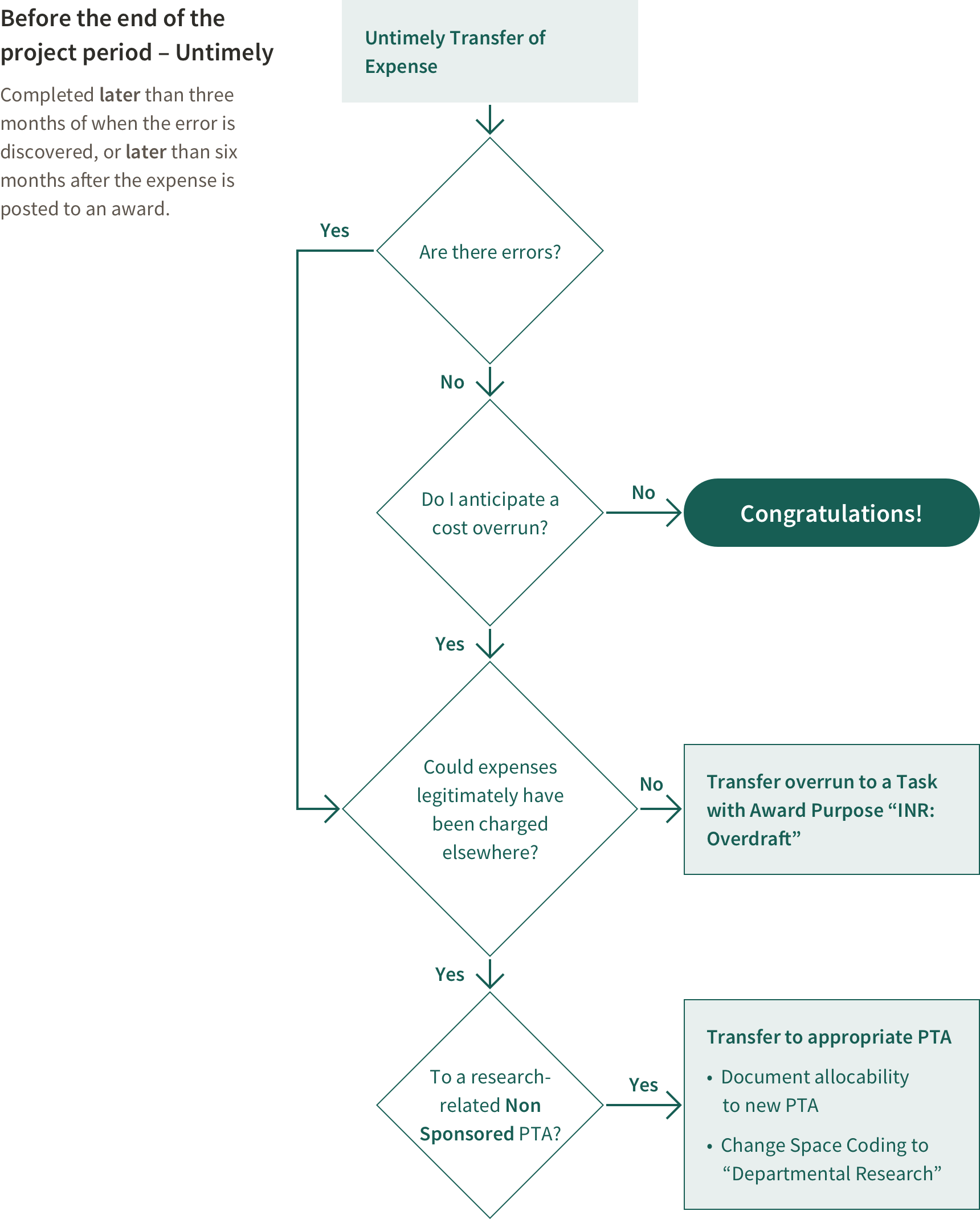
If an error is discovered after the end of the award, a transfer of expense should be made by removing the expense prior to award closeout.
If after the end date of an award an expense is determined to be unallowable to the project (but did benefit the project), the expense must be transferred to a Cost Sharing PTA for accounting purposes, although it cannot be counted towards a Cost Sharing commitment.
Documentation
The documentation of a cost transfer made after a project end date will be closely scrutinized. In addition to including all the necessary elements of a cost transfer justification large cost transfers that exceed $10K or 10% of the award, and transfers within the first or last 90 days of a project, and transfers that do not meet the timeliness criteria receive additional central review. For the transfer of all non-salary charges subject to the above criteria, a PDF of the general ledger** showing the expenditure(s) requesting to be moved MUST be attached to the cost transfer transaction by the originator. Attaching detailed documentation for these transfers will facilitate their timely review by the Office of Sponsored Research (OSR).
Examples
These charges are for effort expended before [insert Project Y’s end date] and they are appropriate per the Project Y award agreement.
These charges could not be processed in a timely manner because PI Smith was traveling in Mongolia and was not available to review and approve the charge.
If a Project Ends in Overdraft
An overdraft exists if after the end date of an award expenses exceed funding.
If the award is in overdraft at the end of the project period, remove the overdraft from the award according to rules outlined in Stanford Policy. Federal regulations state an overdraft is unallowable on any other sponsored project, therefore unless there was an error, the overdraft must be treated like cost sharing. This must be done in a timely manner. Expenses removed as a result of an overdraft should have been incurred during the last 6 months of the project.
-
Do it in a timely manner
-
If total overdraft is less than $500, transfer lump sum (net of indirect costs) Expenditure type 56135 (which allows the Cost and Management Analysis group to segregate these costs for purposes of indirect cost calculation)
-
If the overdraft is greater than $500 dollars the overdraft is transferred to a cost sharing PTA.
-
Explain the reason for the transfer. For example: Charges are legitimate project expenses, but funds were inadequate. This is accounted for in the same manner as cost sharing.
Created: 03.11.2021
Updated: 04.24.2024