NIH Investigators, please review this new NIH requirement for providing copies of agreement(s) related to employment or certain appointments with non-US entities.
For priority review, please upload your Agreements in OPACS by Friday, December 17, 2021. All NIH senior/key personnel should complete the upload of all existing agreements by January 7, 2022
NIH Policy
Effective January 25, 2022, all Stanford University investigators supported by an NIH grant as senior/key personnel must comply with the new NIH Other Support disclosure requirements. This includes providing copies of agreements related to employment or appointments at non-US entities, for example:
- copies of contracts or letters related to an appointment or position with a non-US entity
- copies of grants, contracts, or other sponsored agreements awarded to the non-US entity where you are named as the PI or another role through your appointment at that entity
- copies of agreements that include “in-kind” support related to the appointment, including access to space, facilities, personnel, funding, or other resources available to you in support of your research endeavors (no matter where the research takes place)
- Note that NIH requires that you report all current appointments in the biosketch even if the appointment does not lead to an agreement to be uploaded as part of your Other Support.
Decision Tree for upload of international/foreign agreements to be used as supporting documentation for NIH Other Support
Do I need to upload an international agreement into OPACS for review to allow for future supporting documentation for other support on NIH grant proposals and progress reports (RPPR)?
Note: International agreements negotiated through the Industrial Contracts Office (ICO), the Office of Sponsored Research (OSR), the Office of Technology Licensing (OTL) or the Research Management Group (RMG) do not need to be uploaded as these were institutionally endorsed through Stanford University.
- Question 1: Are you identified as a PI or Key Personnel on an NIH grant application or award?
-
- If No, stop here.
- Note: If you are identified as a Project Director or mentor/advisor on a training grant and have no other NIH support, you should answer no.
- If Yes, go to Question 2.
- If No, stop here.
- Question 2: Do you hold a position through employment with a foreign/international entity or hold an appointment, position or affiliation with an international/foreign entity? (see Definition: Scientific, Research, or Academic Appointments below)
-
- If No, stop here.
- If Yes, go to Question 3.
- Question 3: Does the agreement associated with your appointment, position, or employment with the international/foreign entity provide support to or is it related to your research endeavors, regardless of where the research takes place?
-
- If No, stop here.
- If Yes, go to Question 4.
- Question 4: Is or will the agreement, appointment, or position be active during the same time as your participation in the NIH awarded activity?
-
- If No, stop here.
- If Yes, (1) you will need to disclose the entity in OPACS (if you have not already disclosed it); (2) mark either “appointment or employment” as the type of outside activity with the foreign/international entity (3) upload a copy of the agreement(s) associated with that foreign/international entity; and (4) include a copy of the agreement in your other support form during the NIH Just-In-Time process and when filing progress reports (RPPR) after January 25, 2022.
Note: International Appointments, Affiliations, and Positions that do not involve research support or are not related to your research endeavors may still need to be reported in OPACS as part of your COI/COC disclosure to Stanford and in the biosketch section of your application but may not require a copy of the actual agreement to be uploaded. The review team will return any agreement(s) to you that DOES NOT need to be uploaded with your NIH Other Support.
Definition: Scientific, Research, or Academic Appointments at other institutions
Report appointments that you have, regardless of the title of the appointment, where you are directly involved in any of the following activities:
- the design, conduct, or reporting of academic activities either independently or in support of someone at the other institution;
- providing advice for establishing research programs or laboratories;
- the preparation of proposals, progress, and final reports;
- assisting in the guidance or training of postdoctoral scholars, graduate, and undergraduate students;
- participation in a foreign talent scholar program (If you need help with identifying talent scholar programs, please see information on Academic Integrity and Undue Foreign Interference;
- or helping other researchers in attaining the goals of research projects.
Under this appointment, you could be the co-author or sole author of scholarly papers, including reporting of research results.
Examples of appointments that do not need to be disclosed:
- an honorary title was given to you for presenting or giving a talk on publicly disclosed information (non-proprietary), and there is no further involvement with the host institution
- courtesy appointments or affiliations given to you where there are no expectations for you to teach, perform research, or mentor individuals.
- Serving on a one-time or ad-hoc dissertation or review panels for another institution
- Serving on boards of directors where there is no relevance to your Stanford research endeavors.
Relevant NIH FAQs on Other Support and Appointments/Positions
Do outside consulting activities have to be reported in Other Support?
Consulting activities that involve research and fall outside of an individual’s appointment, separate from the institution’s agreement, must be disclosed as Other Support.
Yes. While the researcher is not receiving monetary compensation, the lab space, materials, and staff are resources made available to them in support of and/or related to their research efforts. Other payments, such as travel or living expenses must also be reported. As outlined in NOT-OD-19-114 this appointment must be reported as Other Support. NIH requires applicants to list all positions and scientific appointments both domestic and foreign held by senior/key personnel that are relevant to an application including affiliations with foreign entities or governments. This includes titled academic, professional, or institutional appointments whether or not remuneration is received, and whether full-time, part-time, or voluntary (including adjunct, visiting, or honorary).
Yes. Other Support includes domestic research collaborations that directly benefit the researcher’s research endeavors. [Stanford note: if the collaboration agreement was signed through Stanford, you do not need to upload a copy of your international agreement – just note the other support in the form. If, however, you signed a collaboration agreement directly with the foreign entity, the agreement will need to be uploaded into OPACS and included as supporting documentation in your other support page].
Yes. Available resources in support of and/or related to an investigator’s research endeavors should be disclosed even if they relate to work that is performed outside of a researcher’s appointment period.
What should I do if I’m not sure if something needs to be included as Other Support?
In the interest of full transparency, recipients should err on the side of disclosure. Researchers should consult with their institutional officials for guidance to ensure compliance with institutional and NIH policies. NIH requires complete and accurate reporting of all sources of research support, financial interests and affiliations, both foreign and domestic.
What types of foreign contracts are recipients required to submit with Other Support?
For Other Support submissions that include international activities and resources, NIH requires researchers to provide:
- international contracts,
- international grants, or
- any other international agreements specific to senior/key-personnel foreign appointments, affiliations, and/or employment with a foreign institution.
Copies must be provided as part of the PDF following the Other Support format page.
[Note: Agreements negotiated and endorsed by an institutional official at Stanford University do not need to be provided.]
What does NIH evaluate when reviewing Other Support submissions?
NIH scientific program and grants management staff review Other Support information to ensure that:
- All resources, domestic or foreign, directly supporting the individual’s research endeavors have been reported
- Sufficient levels of effort are committed to the project
- There is no scientific, budgetary, or commitment overlap
- Only funds necessary to the approved project are included in the award
- Any foreign resources that meet the definition of a foreign component have received appropriate prior approval.
See NIH’s matrix for a summary of items that should be included in your biosketch or other support form at the JIT and RPPR stages.
Stanford FAQs for External Agreements
- 1. How can I get my foreign language agreement translated into English before I submit to Stanford?
-
a. You can use a translation service provider.
b. You can use Google Translate or other services deemed to provide accurate automated translations.
c. If you are bilingual and can accurately translate from a foreign language to English, this may be an option.
Submit only the English version of the documents in PDF format.
- 2. What format should I use to upload documents to OPACS?
-
a. Please only upload PDF versions of your documents.
NIH FAQs related to Foreign Contracts
NIH requires researchers to provide foreign contracts, grants or any other agreements specific to senior/key-personnel foreign appointments, affiliations, and/or employment with a foreign institution. Copies must be provided as part of the PDF following the Other Support format page.
2. Does this include employment contracts for fellows or students working in the lab?
No. Personal service contracts for lab staff do not need to be provided. Only contracts, grants and any other agreements specific to the PD/PI or other senior/key personnel’s appointments and employment are required. However, NIH continues to reserve the right to request additional information, as necessary.
Yes. The requirement to provide foreign contracts, grants or any other agreements applies to all Other Support information submitted on or after May 25, 2021. In the Research Performance Progress Report (RPPR) for ongoing grants, recipients must provide copies of foreign contracts, grants or any other agreements specific to senior/key-personnel foreign appointments and/or employment with a foreign institution for all foreign activities and resources that are reported in Other Support.
4. How will NIH review foreign contracts, grants or any other agreements that are provided?
NIH requires the submission of foreign contracts, grants or any other agreements specific to senior/key-personnel foreign appointments and/or employment with a foreign institution, as supporting documentation to the Other Support submission. NIH will review contracts, grants or any other agreements to confirm that all information provided in the Other Support submission is accurate and complete.
No. The researcher’s employment contract with the applicant organization does not need to be provided. Contracts, grants or any other agreements with outside entities, such as other foreign organizations, must be provided.
Yes. NIH will accept machine-read translations.
7. Are the costs of translating foreign contracts allowable on NIH grants as a direct cost?
The costs associated with translations of foreign contracts for inclusion in Other Support submissions are not typically allocable to a specific NIH grant project and are therefore not allowable as a direct cost. NIH recommends that institutions consider including such costs in their negotiations for indirect cost reimbursement.
NIH needs access to all information necessary to make an informed judgement about possible scientific, budgetary, or commitment overlap and to make an informed judgment about management of possible significant financial conflicts of interest. NIH handles confidential and sensitive information in accordance with our cybersecurity requirements and requires submission of complete documentation.
Supporting documentation is required for active and pending support specific to senior/key-personnel foreign appointments, affiliations, and/or employment with a foreign institution.
Yes. If the individual is performing activities under any active contract, grant or other agreement, it must be provided as supporting documentation.
Stanford Process
In preparation for this new requirement, OPACS has been enhanced to allow investigators to submit copies of these external agreements for Stanford review to ensure compliance with Stanford policy, including:
- Identification and management of any overlap between the outside activity and the Investigator’s Stanford responsibilities, per RPH 4.1 – Faculty Policy on Conflict of Commitment and Interest, and RPH 4.4 – Conflict of Commitment and Interest for Academic Staff and Other Teaching Staff
- Identification and management of any overlap between the outside activity and the Investigators sponsored awards, per RPH 4.2 – PHS and NSF Requirements
- That the time commitment complies with RPH 4.3 – Consulting and Outside Professional Activities, and
- That appropriate prior approval has been received for appointments that fall under Faculty Handbook 2.7.7 – Appointments at Other Institutions
Once reviewed, the Agreements will be available for submission with your Other Support. Note: The agreements will be made available to your department research administrator to prepare the Other Support package, and your Institutional Official in RMG and OSR for submission to the NIH as required.
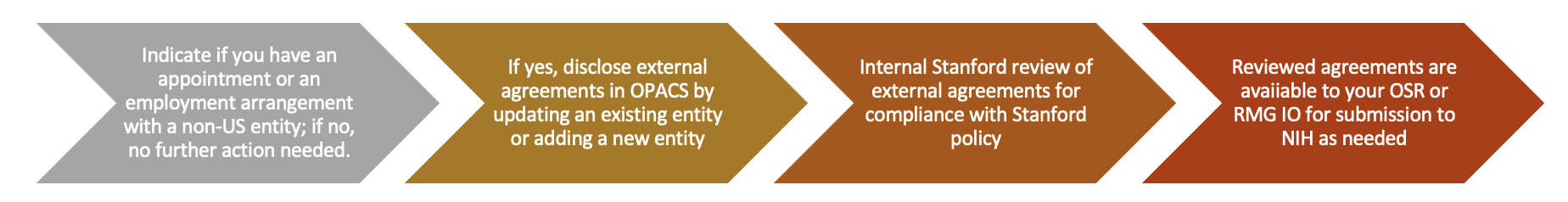
Stanford is Taking a Phased Approach
Catch Up Phase
Collect existing agreements in OPACS related to employment or appointment at a non-US entity and active grants and contracts through your appointment at the non-US entity as described above.
All investigators named as senior/key personnel on NIH pending proposals or active awards will receive an email in early December asking if they have an appointment with a non-US entity that must be reported to the NIH under these new requirements.
- If No – the Investigator registers a “no” response in the email, and no additional information will be needed.
- If Yes – the Investigator will follow the instructions below to provide copies of all relevant agreements in OPACS. Note, agreements must be in English.
Reminder: all agreements must be reviewed by Stanford before submission to the NIH. Before submission, your Institutional Official in RMG or OSR will verify that Stanford has reviewed any agreement included in an investigator’s Other Support via this process.
For priority review, please upload your Agreements in OPACS by Monday, December 13, 2021. All NIH senior/key personnel must complete the upload of all existing agreements by January 7, 2022
Going Forward Phase beginning December 2021
For new proposals and project updates for new awards, as part of the SeRA proposal certification process, investigators will be reminded to update their OPACS information and disclose and upload external relationships and new/amended agreements.
Additional Information
Reminder: Stanford policies require disclosure of new entities and updates to existing entities in OPACS within 30 days of a new activity or when your relationship with an existing entity changes.
Update OPACS and upload external agreement amendments or changes to current agreements within 30 days of a change. In addition, investigators and Stanford are required to provide timely updates to NIH for significant changes in Other Support, including new external agreements or revisions to previously reported external agreements.
Upload external agreements once. External agreements will be available to your Institutional Official in OSR or RMG to submit to NIH as needed for any NIH-funded project where you are a named Investigator.
Confidentiality
Once reviewed, the agreements will be available for submission with your Other Support documents to NIH. Note: your Institutional Official in RMG and OSR will also have access to these agreements and individuals who assist you with including the agreements in your NIH proposal will also have access to upload these agreements. Once submitted to NIH, individuals at NIH will also see the contents of these agreements and NIH has stated that they do not want portions of the agreements redacted. If the agreement contains a confidentiality clause that does not allow this level of dissemination, you may need to work with the outside entity to modify the current terms in order to be compliant with NIH’s new disclosure requirement.
Please submit questions by submitting a ticket and we’ll help you make the appropriate disclosures.
Resources
NIH FAQs regarding external agreements (See section I. Other Support, D. Foreign Contracts)
OSR page on disclosures on NIH and other federally funded projects
RMG page on disclosures on NIH and other federally funded projects
NIH Notice OD-21-073 – March 2021